【News】Medical HAL Single Joint Type receives medical device approval from the regulator of Taiwan
CYBERDYNE Inc. (Tsukuba, Ibaraki, Japan, President & CEO: Yoshiyuki Sankai) announced today its application for a medical device approval for Medical HAL Single Joint Type, a Wearable CyborgTM that improves physical functions, has been approved by the Taiwan Food and Drug Administration (TFDA).
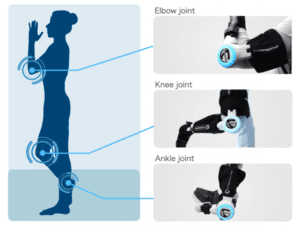
The Company has obtained medical device approvals in Japan, the USA, and the EU for this device. The device is also approved in the Asia-pacific region, and it is being disseminated in each country. In this region, the approval in Taiwan follows those in Malaysia, Thailand, and Australia. In collaboration with CHC Healthcare Group (承業生醫企業集團), a major medical device distribution company and a hospital affiliated to CHC Healthcare Group, Cybernics Treatment Centre are now in operation (Please refer to the press release on March 29, 2021). The Company will continue to disseminate Cybernics Treatment with HAL Lower Limb Type and Single Joint Type with its partners to contribute to realizing a society of health and longevity where all people can enjoy healthy and affluent lives.
Reference)
Medical HAL Single Joint Type receives medical device approval from the Australian regulator (September 2, 2021)
https://www.cyberdyne.jp/english/company/PressReleases_detail.html?id=11395
CYBERDYNE listed Medical HAL Single Joint Type to U.S. FDA as Class I Medical Device (August 30, 2021)
https://www.cyberdyne.jp/english/company/PressReleases_detail.html?id=11345
Grand Opening of Cybernics Treatment Center in Taiwan (March 29, 2021)
https://www.cyberdyne.jp/english/company/PressReleases_detail.html?id=10664
HAL Single Joint Type obtained certification as a medical device for Japan, applied for insurance coverage (July 13, 2020)
https://www.cyberdyne.jp/wp_uploads/2020/07/20200713_PR_eng.pdf
HAL Single Joint Type obtained conformity certification as a medical device for EU (October 8, 2019)
https://www.cyberdyne.jp/wp_uploads/2019/10/20191008_PR_Single-Joint_ENG.pdf